
Results showed that the use of an ABGS allowed for a settling velocity of 3.4 m/s for Zn/Pb precipitates from a real wastewater. In the second case, the use of an abiological granular sludge (ABGS) was used to improve the aggregation and settling of sulfide precipitates. 
This result indicates that the aggregation of particles could increase after applying a magnetic field, but no PSD or settling test were performed. In the first case, the application of a magnetic field of 2T for 40 min increased the zeta potential from −40 mV to 16.5 mV. The use of a magnetic field to modify the zeta potential of particles, the use of an abiological granular sludge, and the use of Ultraviolet (UV) light irradiation to improve the aggregation have been experiments conducted to enhance the settling rate of particles. Studies to optimize the solid–liquid separation in metal sulfide precipitation processes are still emerging, although there are a few interesting examples which could address new research trends. In this regard, there are critical studies that showed the fragility of the overall process of recovery with respect to a correct control of the clarifier, losing even 40% of copper recovery in this stage. Furthermore, there are several opportunities to improve these results, such as the size reduction, minimization of solid losses in the overflow, and the process control. Although these industrial applications have reported to be successful, they are still limited to the recovery of a few metals, mainly copper. At an industrial scale, the current equipment used to separate precipitates are conventional gravitational clarifiers, as used, for example, in the SART process and for the treatment of AMD. This situation imposes high variability of the particle size distribution (PSD) that results from the reaction stage, increasing the uncertainty of solid–liquid separation performance. 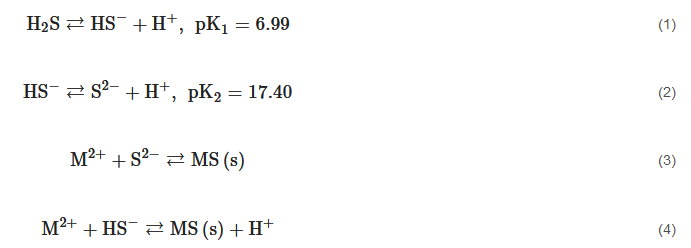
As discussed in the previous section, the particle behavior of sulfide precipitates is determined by its hydrophilicity/hydrophobicity capacity, the ions contained in solution, the sulfide concentration, and pH.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
