
People who fail to respond (anti-Hbs antibody level below 10 mIU/ml) should be tested to exclude current or past hepatitis B infection, and given a repeat course of three vaccinations, followed by further retesting 1–4 months after the second course. An antibody level between 10 and 100 mIU/ml is considered a poor response, and these people should receive a single booster vaccination at this time, but do not need further retesting. Īn anti-Hbs antibody level above 100 mIU/ml is deemed adequate, and occurs in about 85–90% of individuals. Testing to confirm successful immunization or sustained immunity is not necessary or recommended for most people, but is recommended for infants born to a mother who tests positive for HBsAg or whose HBsAg status is not known for healthcare and public safety workers for immunocompromised people such as haemodialysis patients, HIV patients, haematopoietic stem cell transplant recipients, or people receiving chemotherapy and for sexual partners of HBsAg-positive people. Studies have found that that immune memory against HepB is sustained for at least 30 years after vaccination, and protects against clinical disease and chronic HepB infection, even in cases where anti-hepatitis B surface antigen (anti-Hbs) levels decline below detectable levels. Hepatitis B immunoglobulin before birth has not been well studied. The vaccine during pregnancy is not considered to be valuable in protecting babies of the infected mothers. 
The combination is superior for protecting these infants. Hepatitis B vaccination, hepatitis B immunoglobulin, and the combination of hepatitis B vaccine plus hepatitis B immunoglobulin, all are considered as preventive for babies born to mothers infected with hepatitis B virus (HBV). and is recommended by the Advisory Committee on Immunization Practices (ACIP). A pentavalent vaccine combining vaccines against diphtheria, tetanus, pertussis, hepatitis B, and poliomyelitis is approved in the U.S. There is not yet sufficient evidence on how effective this pentavalent vaccine is in relation to the individual vaccines. The World Health Organization (WHO) recommends a pentavalent vaccine, combining vaccines against diphtheria, tetanus, pertussis and Haemophilus influenzae type B with the vaccine against hepatitis B. The US Centers for Disease Control and Prevention (CDC) issued recommendations for vaccination against hepatitis B among patients with diabetes mellitus. Both types of the vaccine, the plasma-derived vaccine (PDV) and recombinant vaccine (RV), seems to be able to elicit similar protective anti-HBs levels. In many areas, vaccination against hepatitis B is also required for all health-care and laboratory staff. A similar situation is in operation in Ireland. In the UK, the vaccine is offered to men who have sex with men (MSM), usually as part of a sexual health check-up. This was reported in Taiwan where the implementation of a nationwide hepatitis B vaccination program in 1984 was associated with a decline in the incidence of childhood hepatocellular carcinoma. In countries with high rates of hepatitis B infection, vaccination of newborns has not only reduced the risk of infection, but has also led to a marked reduction in liver cancer. Many countries routinely vaccinate infants against hepatitis B. In the United States vaccination is recommended for nearly all babies at birth. Both versions were developed by Maurice Hilleman and his team. It is on the World Health Organization's List of Essential Medicines. A recombinant version came to market in 1986. The first hepatitis B vaccine was approved in the United States in 1981. 
They are available both by themselves and in combination with other vaccines. Hepatitis B vaccines are produced with recombinant DNA techniques and contain immunologic adjuvant. It has not been linked to Guillain–Barré syndrome. It is safe for use during pregnancy or while breastfeeding. Serious side effects from the hepatitis B vaccine are very uncommon. The vaccine is given by injection into a muscle. In those who have been exposed to the hepatitis B virus (HBV) but not immunized, hepatitis B immune globulin should be given in addition to the vaccine. Additional doses may be needed in people with poor immune function but are not necessary for most people. īlood testing to verify that the vaccine has worked is recommended in those at high risk. In healthy people, routine immunization results in more than 95% of people being protected. It is also recommended that health-care workers be vaccinated. 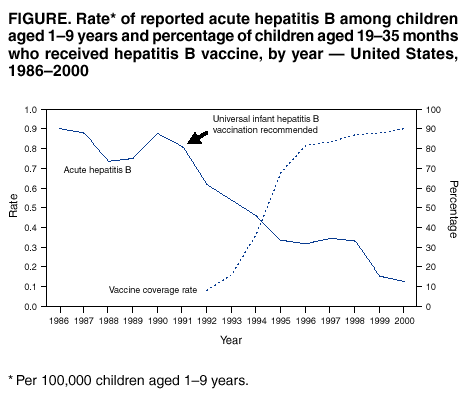
This includes those with poor immune function such as from HIV/AIDS and those born premature. The first dose is recommended within 24 hours of birth with either two or three more doses given after that. Hepatitis B vaccine is a vaccine that prevents hepatitis B.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
